|
The Ideal Gas Law can be expressed with the Individual Gas Constant. The Ideal Gas Law - or Perfect Gas Law - relates pressure, temperature, and volume of an ideal or perfect gas. 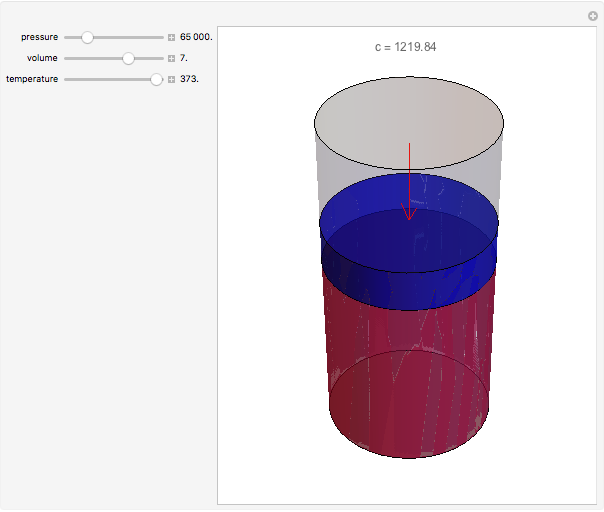
The Ideal Gas Law and the Individual Gas Constant - R One mole of an ideal gas at STP occupies 22.4 liters.R u = universal gas constant, = 8.3145 = 0.08206 = 62.37 įor a given quantity of gas, both n and R u are constant, and Equation (1) can be modified toĮxpressing the relationship between different states for the given quantity of the gas. N = is the number of moles of the gas present The Universal Gas Constant, R u is independent of the particular gas and is the same for all "perfect" gases, and is included in of The Ideal Gas Law: 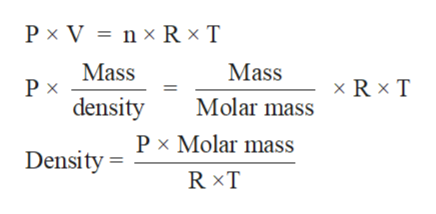 
In a perfect or ideal gas the correlations between pressure, volume, temperature and quantity of gas can be expressed by the Ideal Gas Law.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
